Engineered Human Therapies
Designing Programmable Cancer Therapies with Synthetic Gene Circuits
Synthetic gene circuits make cancers more visible to immunotherapies while minimizing their toxicity and adverse effects
[DALL-E]
Cancers are incredibly complex, primarily because there is a lot of crosstalk between cancer, host cells, and the environment. This complexity often limits the success of small molecule or antibody-based therapeutics in cancer treatment. These therapeutics usually target a single antigen and are better suited to diseases with simple mechanisms.
“There are multiple targets you have to address for most cancers,” said Tim Lu, CEO of Senti Bio, a biotech company based in South San Francisco. Today, it’s pretty easy to modify a cell to express a protein that will make an antigen bind to it. “Put in a combination of DNA constructs with a set of rules about how and when they need to be activated, and that is a synthetic genetic circuit,” said Rogelio Hernandez-Lopez, assistant professor of bioengineering and genetics at Stanford University.
How cells behave depends on interactions between different cells and between subcellular components within cells. With synthetic gene circuits, biologists build upon these interactions to create cells with custom functions, such as programmable therapies. Since gene circuit components are modular, like those in electronic circuits, these therapies can be adapted to different clinical needs.
The Logic of Programmable Therapies
Generally, synthetic gene circuits leverage the transcription machinery to manipulate the expression of particular genes. This is called transcriptional logic. Hernandez-Lopez provided an example of the if-then logic that’s possible with these circuits. “If there are a lot of tumor-specific antigens expressed on a cell, then express a particular protein that triggers immune cells to kill the cell,” said Hernandez-Lopez.
In cancer treatment, “the key challenge is to distinguish cancer cells from healthy cells,” said Lu. With synthetic gene circuits, researchers can devise mechanisms to discriminate cancer cells from normal tissue based on the antigens expressed on the surface of a cell. And it’s just not limited to the expression of a particular antigen, these circuits can recognize and integrate information from multiple antigens.
This is where logic-gated gene circuits come in. Think of a gene circuit that activates a cancer-killing immune cell only when it recognizes two or more antigens (and a Boolean relationship between them). Moreover, “new classes of synthetic receptors allow you to control when to express a particular payload,” said Hernandez-Lopez.
Synthetic gene circuits don’t just need to look for antigens on cancer cells. “Instead of just looking for one target on cancer cells that triggers killing, what if you can find a marker of healthy cells that prevents your drug from killing them? That additional information can get you dramatically greater specificity against cancer,” said Lu. This makes cancer cells more visible to cancer therapies, limiting off-target effects.
As biologists learn more about cancer’s interactions with its environment, it opens up new possibilities for designing synthetic gene circuits that target cancer cells. For example, Hernandez-Lopez’s research group is engineering cell-cell communication between T cells and other cells of the immune system, such as antigen-presenting cells or neutrophils, that deal with the tumor microenvironment.
Immune Cell Engineering
In recent years, engineered immune cells have become a promising approach for cancer treatment. These are therapies where immune cells derived from the patient or a donor (for off-the-shelf therapies) are modified to contain a synthetic gene circuit.
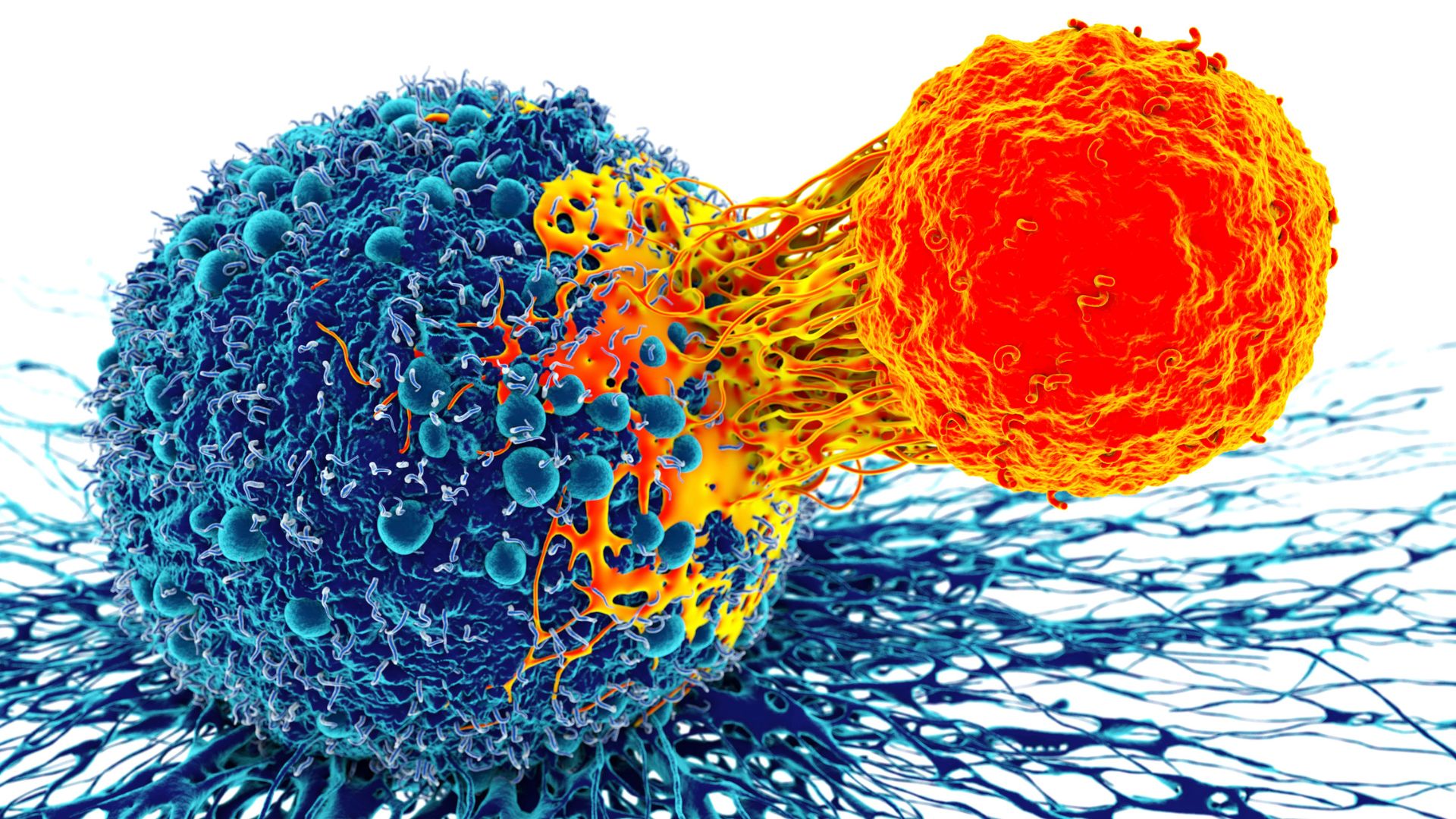
Most notable among these cell-based immunotherapies are CAR-T therapies, wherein engineered T cells express chimeric antigen receptors (CAR) on their surface. Since 2017, CAR-T therapies have been approved for B cell malignancies and multiple melanomas, with many others under trial.
The receptors on CAR cells are called chimeric because they have multiple parts. “A part [of the receptor] will guide the T cells against a particular protein expressed on the surface of cancer cells, and then signaling domains allow the T cell to activate killing, proliferation, and other functions,” explained Hernandez-Lopez. These can be logic-gated as well. For example, CAR-T cells can be designed to activate only when they receive signals in the tumor microenvironment.
CAR therapies built on other immune cell types are also under investigation. For example, Senti Bio engineers natural killer (NK) cells to create CAR-NK therapies. While no CAR-NK drugs have been approved to date, they offer promise as they have lesser toxicity and easier manufacturing for off-the-shelf use.
SENTI-202, Senti Bio’s lead drug candidate, uses a combination of OR and NOT logic gates to treat acute myeloid leukemia (AML). It attacks a cell only if its surface has at least one of two particular markers overexpressed in blast cells and leukemic stem cells of acute myeloid leukemia but does not express a marker seen on healthy hematopoietic stem cells. The drug is expected to enter phase 1 clinical trials in 2024.
Unlike CAR-T therapies, which usually have a transcriptional logic, Senti Bio’s CAR-NK therapies rely on protein signaling cascades. Compared to transcriptional logic, protein logic is faster and enables more precise cellular responses.
mRNA Immunotherapies
mRNA therapeutics have received significant attention in recent years, particularly since the first mRNA-based COVID-19 vaccines. For cancers, mRNA vaccines can quickly be developed for patient-specific antigens for personalized cancer treatment. With synthetic gene circuits, scientists can combine the benefits of mRNA with the broad applicability of off-the-shelf CAR therapies.
A significant benefit of mRNA immunotherapies is the improved safety. Viral vectors used for genetically modifying immune cells can have adverse effects. mRNA therapies are delivered via non-viral vectors, and the mRNA is transient. Moreover, mRNA doesn't integrate into the genome. If the rapid development of mRNA-based COVID vaccines is any hint, mRNA immunotherapies might lead to faster development of cancer therapies.
Another benefit of mRNA immunotherapies compared to CAR therapies is their transfection efficiency. DNA encoding for the CARs needs to get inside the nucleus of T cells. On the contrary, with mRNA, “once you get it inside a cell, it can translate into protein,” said Tasuku Kitada, co-founder and head of R&D at Boston biotech Strand Therapeutics.
The company develops off-the-shelf programmable mRNA therapies for hematological malignancies and solid tumors. Kitada added, "Strand’s synthetic mRNA circuits are agnostic to tumor type and activate the patient's immune system from within the tumor microenvironment.” The company works on self-replicating and circular mRNA modalities. Unlike linear mRNA, these mRNA types provide more durable protein expression and, hence, sustained therapeutic benefit.
However, Kitada stressed that the fact that mRNA can easily enter cells and express protein in any cell type is a double-edged sword. If a very potent mechanism is encoded in the RNA, it can induce a lot of toxicity. “Regardless of the RNA modality, Strand programs these RNAs so that you can specifically express proteins in cancer cells, immune cells, or whatever cell types that you're interested in,” said Kitada.
This precise spatial control underlies the growing traction of mRNA immunotherapies. As synthetic biologists expand on the toolkit of gene circuits, it would allow the development of more sophisticated synthetic gene circuit architectures.
This could reduce the toxicity of engineered immune cells while making hard-to-treat cancers, such as solid tumors, more amenable. New mRNA immunotherapies beyond mRNA vaccines will serve unmet clinical needs in cancers, as well as other indications such as autoimmune diseases and cardiovascular diseases.


















