Engineered Human Therapies
Phages and Synthetic Biology Team Up to Tackle Antimicrobial Resistance
Engineered phages offer several promising approaches to treating antimicrobial-resistant infections, yet manufacturing them is tricky

Science Photo Library (Canva)
The first antibiotic was discovered in 1928 by Alexander Fleming. In the decades that followed, new antibiotics were discovered rapidly and quickly modified to make them more effective. But, with the increasing use of antibiotics, it’s become more challenging to identify new antibiotics that are effective against “superbugs” or bacteria resistant to multiple classes of antibiotics. No new classes of antibiotics have been brought to market since the 1980s.
While one could “simply” develop new classes of antibiotics, there are many reasons why companies aren’t doing this. Some of it is economics: it takes over a decade for an antibiotic candidate to progress to clinical trials, it’s costly, and not all drug candidates make it to market. And unfortunately, bacteria could become resistant to the drug in just a few years.
An alternative to antibiotics? Phage therapy and engineered phage.
Treating Bacterial Infections Without Antibiotics
The idea is simple: a bacteriophage, a virus that infects bacteria, can be used to treat bacterial infections. They enter the bacteria, use them to replicate, kill them, and then emerge to infect other bacteria. Phages are specific only to a subset of bacteria, and this feature is based on structures on the bacterial cell surface, like lipopolysaccharides, teichoic acids, proteins, pili, or flagella, that act as signals for phage attachment. If the bacteria do not possess the correct signal, the phage can’t infect.
Phage therapy has been used for over a hundred years to treat bacterial infections. However, this type of therapy is not routinely used in Western countries as antibiotics became widely used and is now only available in cases of “compassionate use” in the United States in which immediately life-threatening illnesses are faced with no alternative approved options.
Cases like this require quick action to pair the correct phages to the bacterial infection. And this is exactly what organizations such as Phage Directory aim to do. “Phage Directory coordinates treatment of antibiotic-resistant infections by sourcing phages from a community of researchers across 80 countries,” says Jessica Sacher, a postdoctoral research scientist at Westmead Institute for Medical Research and cofounder of Phage Directory. They’re now scaling up to more patients through a partnership with a medical research/hospital network in Australia and plans to expand globally.
“We've saved a 7-year-old's leg from amputation, helped a 2-year-old get phages to treat a liver infection, helped a sea turtle get phages for a shell infection, all through researcher-contributed phages,” says Sacher.
However, phage aren’t necessarily the best bacterial killers. “Often phages in the wild are not as efficient killers as you'd want them to be since it's not in their natural best interest to obliterate their host,” says Sacher. It’s also difficult to control phage replication dynamics, and phages can often carry “genetic baggage like antibiotic resistance cassettes and toxins, and might even worsen an infection,” says Irene Chen, Associate Professor at the Department of Chemical and Biomolecular Engineering at UCLA.
To overcome these challenges, phages need some help. Enter synthetic biology.
Synthetic Biology Optimizes Phages As Alternatives to Antibiotics
“Phages are a robust, biocompatible nanomaterial that we can engineer in different ways. We’ve only just scratched the surface,” says Chen. Chen’s lab examines how phages can be genetically engineered, chemically modified, or conjugated with nanomaterials for diagnostic and therapeutic applications.
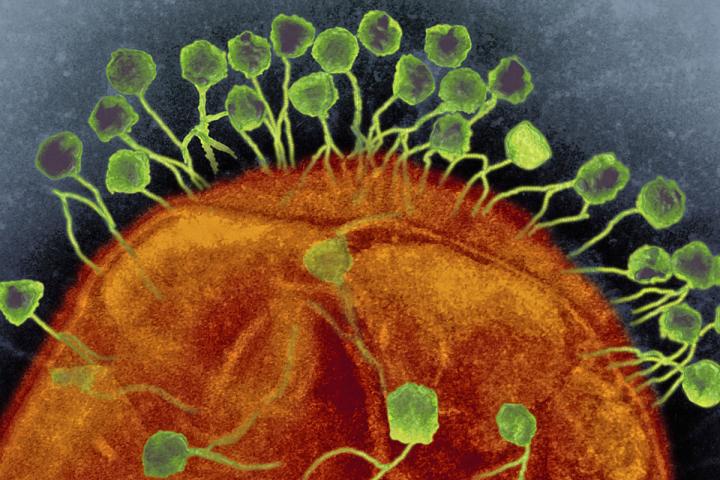
What makes phages great for engineering is their natural ability to tolerate genetic mutations. “Phages engage in an ‘evolutionary arms race’ with bacteria, where both sides are innovating genetically as quickly as possible to resist and overcome each other,” says Chen. “We speculate that this rapid pace of adaptation has selected, over the long run, for protein sequences that are particularly tolerant of genetic changes. We think that maybe phage proteins are also particularly tolerant of our engineering too.”
Some of these approaches include engineering phages as delivery tools that can bring a particular payload to specific bacteria or enhance them to become better bacterial killers.
“Viral vector deliveries of genetic tools of any kind is the future of medicine,” says Paul Garofolo, cofounder and CEO of Locus Biosciences, a clinical-stage biotech company developing phage therapies based on CRISPR-Cas3, which chews back long stretches of DNA. Locus Biosciences is developing multiple phages as precision therapeutics to target E. coli urinary tract infections and infections with P. aeruginosa, S. aureus, and K. pneumoniae. Their CRISPR-enhanced bacteriophage cocktail (crPhage®) to treat urinary tract infections caused by E. coli is the first randomized Phase 2/3 clinical trial of CRISPR-enhanced bacteriophage to treat bacterial infections.
Eligo Biosciences is taking a similar approach. But instead of using Cas3, they’re using Cas9 to target specific sequences, such as those that encode toxins or proteins that make bacteria resistant to antibiotics.
Other companies use phages to deliver other “antimicrobials” to drug-resistant infections. For example, Phico Therapeutics uses phage to deliver a gene encoding for a pan-spectrum antibacterial protein. Once expressed by the bacteria, this protein binds and inactivates bacterial DNA, and the cells can’t replicate. This gives the immune system time to clear the bacteria from the body. This approach has many advantages that reduce the potential for antimicrobial resistance as the antibacterial protein can bind any DNA sequence. Therefore, the therapeutic is still effective despite the bacteria’s effort to circumvent antimicrobials (e.g., mutations or newly acquired DNA).
While the above approaches rely on internalizing phage to release the payload, Chen’s lab is engineering phage-nanomaterial conjugates to target Pseudomonas wound infections without entry into bacteria. In this method, phage particles deliver gold nanorods and zinc to the bacterial infection. When excited by light, the gold nanorods release heat to kill the bacteria, and zinc, which promotes wound healing and inhibits bacterial growth, is released. “We try to re-imagine the phage as a sort of killing machine, delivering a large dose of cytotoxic cargo to the bacterial cell without infecting and replicating,” says Chen. “The tradeoff is that we may need multiple doses (like with antibiotics), but we believe it’s important to avoid relying on natural phage dynamics.”
Theoretically, you can use phage to deliver any payload to bacterial cells. “Whether you're trying to get through a mucin layer, whether you need to contend with biofilm, whether you need to contend with expanding host range, all of these things are engineering problems that phage provide this large and healthy scaffold to be able to do,” says Garofolo.
Aside from delivering payloads, phages can be engineered to bind better and infect specific bacteria. “We've spearheaded being able to engineer enzymatic components onto the outside of the [phage] capsid so that [phage] binding and potentially extension of its natural host range is expanded,” says Garofolo.
Challenges of Manufacturing Phages
While these approaches—phage therapy and engineered phage—offer great starting points to combat antimicrobial-resistant infections, there’s still the challenge in manufacturing phage.

“Manufacturing phages is tricky because most contract manufacturing organizations don't want phages in their facilities for fear they'll contaminate bacterial strains they're making for other clients,” says Sacher.
That’s why Locus developed its own phage manufacturing facility, which handles phage and other viral vectors.
Aside from finding a facility to manufacture phages, phage biology itself brings a lot of challenges. For starters, every phage requires a different manufacturing process. “Formulation of phages is a challenge due to phage-phage differences in adherence to plastics and glass, differences in preferred buffers and salt compositions, and just general differences in stability even for the same storage conditions,” says Sacher. You’d also need a very high titer to ensure enough binding with the bacterial target. “Anytime you're manufacturing a virus at very high titers, you're typically bringing a lot of toxins along for the ride, so you need a fairly advanced filtration and purification process to get those byproducts out,” says Garofolo.
Can Bacteria Develop Resistance to Phage?
While phages can be great alternatives to the looming antimicrobial resistance problem, can they be effective long-term? In other words, can bacteria develop resistance to phages? The answer is yes, but there’s a caveat to their resistance: phages can also evolve, and it’s possible to use a “cocktail” of phages as bacteria are less likely to develop resistance to multiple phages at once. It’s also possible to swap out phages in a phage cocktail, and there’s another important characteristic of phages that differentiate them from antibiotics: phages replicate and can adapt to changing conditions. “As bacteria evolve, the phage cocktails that go after them can evolve,” says Garofolo. “Resistance is completely different when you're not dealing with a small molecule that is fixed.”
Read More
Newletter & More

SynBioBeta
Join the innovators shaping the future with SynBio + AI. From health to ag, materials & more—be part of the revolution.

















