
Metastatic melanoma cells. Credit: Julio C. Valencia, NCI, NIH
Spatialomics is considered by many to be the next frontier in understanding gene expression. Earlier this year, Nature named spatially resolved transcriptomics—also known as spatialomics—the 2020 Method of the Year. This burgeoning new technology is the combination of tissue imaging and single-cell transcriptomic analysis, the full expression profile of messenger RNA (mRNA). The technique creates an entirely new type of scientific information.Spatialomics information can be used to more deeply understand disease, potentially enabling the development of novel treatments. “A disease ultimately comes from molecular defects in the behavior of some cells. What we've been doing up to date is trying to address that defect without knowing how the system itself works,” says Jeffrey Moffitt, assistant professor at Boston Children’s Hospital, and co-developer of the MERFISH technology that has helped to pioneer spatialomics. “Imagine if you were trying to fix an engine without ever knowing the blueprints of the engine—you can be effective, but you’re limited. What spatialomics does is provide a window to determine the blueprint of a disease system, what the parts are, and how they fit together. I can’t imagine a more useful resource in understanding disease,” says Moffitt.
The Third Generation of Transcriptomic Analysis Tools
Gene expression in a cross-section of a mouse brain.Video: VizgenSpatialomics tools are what could be considered the third generation of transcriptome analysis technologies. The transcriptome is the total messenger RNA (mRNA) in a sample, whether at a whole tissue or single-cell level. This reveals all of the genes being expressed at the moment of analysis.Understanding the transcriptome is important because while the human genome has 3 billion base pairs, the majority of genes these base pairs encode for are either not expressed or are only expressed under certain conditions. This information can inform scientists how the genes expressed by, say, a cancer cell differ from a normal cell. Understanding these differences can garner new insight into diseases and pave the way for novel treatment.Spatialomics builds on the advances to two early gene expression research technologies, bulk RNA sequencing and single-cell RNA sequencing. Both of these methods illustrate just how far spatialomics has come:
Bulk RNA Sequencing
Early efforts to understand gene expression focused on analyzing the total mRNA of a tissue sample, also known as “bulk RNA sequencing.” Using this technique a tissue sample is homogenized (i.e., blended up) and its total mRNA is analyzed to show the average gene expression levels across the entire sample. However, this does not provide details about what the transcriptome looks like on the single-cell level. In many diseases such as cancer, the genetic profile of individual cells in a tumor is different. Without data for each cell, researchers cannot understand the full genetic profile of a tumor or other disease types.
Single-cell RNA sequencing
The next-generation technology in this field, single-cell RNA sequencing (scRNA-seq), enabled researchers to understand the transcriptome of a single cell in a dissociated tissue—a tissue broken apart into its cellular components. This was a pioneering approach, but it did not provide spatial information, which is important for three primary reasons. First, the way one cell transcribes its genes (copies DNA to RNA) affects the way it signals to its neighbor. As this process repeats, each cell’s signal creates a chain of information in a tissue. Second, complex tissues, such as the brain, liver, kidney, and other major organs are not homologous. There are differences between the genes transcribed by the various groups of cells in these tissues. These differences create the distinctions in cellular function needed to maintain a working organ. Third, there are some diseases for which RNA is localized differently than it is in healthy cells.The ability to see where and when genes express could provide a crucial understanding of why and how diseases arise. It could also create a stronger foundation for tissue engineering, from skin grafts to synthetic hearts and kidneys.
New Technology Solves Old Problems
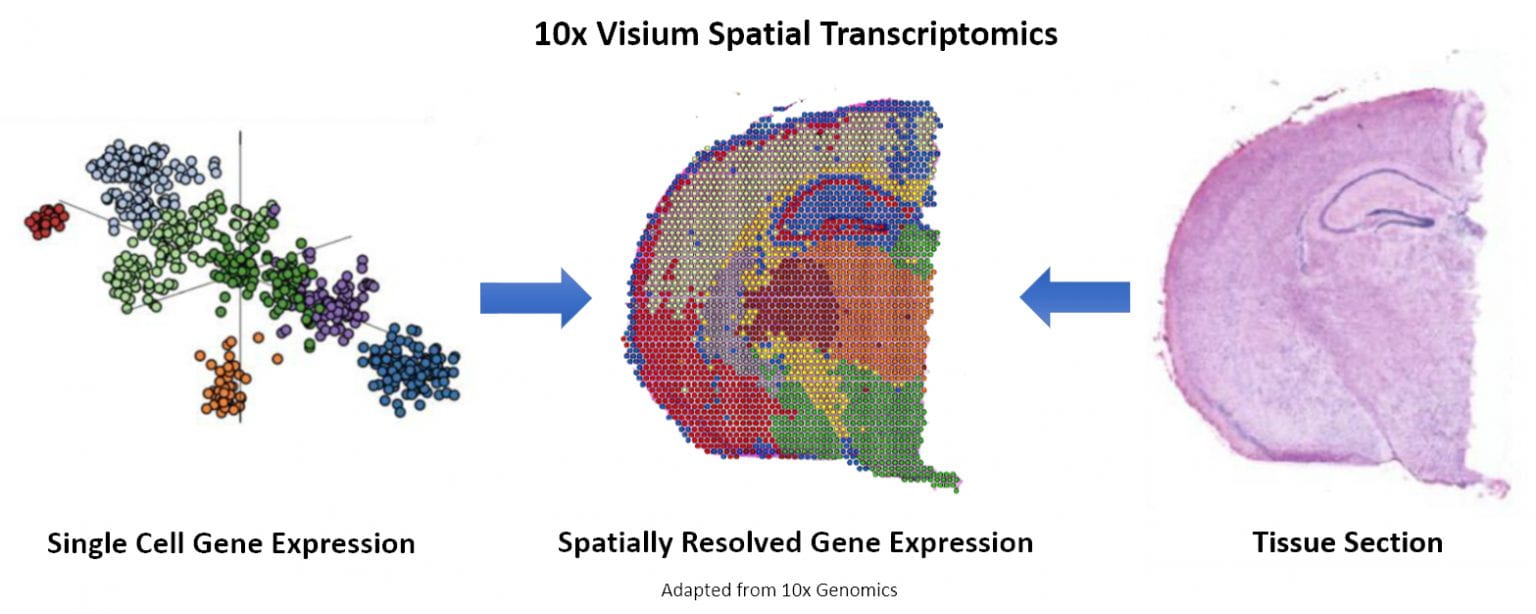
10X Genomics platform, Visium, visually maps gene expression in a tissue sample. Image: 10X GenomicsSpatial single-cell transcriptomics resolves the drawbacks of older bulk RNA and scRNA-seq technologies. By combining imaging and single-cell RNA sequencing, researchers can map where particular transcripts are expressed within a tissue. This can not only reveal the “where” of gene expression but also indicate the context of how individual cells function within that tissue. https://www.nature.com/articles/s41592-020-01033-y “Spatial transcriptomics enables the resolution we need to make connections and refine our understanding of how cells interact within a tissue,” says David Walt, a pioneer in microwell arrays and single molecules, scientific founder of Illumina Inc., and co-founder of Vizgen. “Earlier methods give us a picture of tissue heterogeneity, but achieving cellular and sub-cellular resolution is needed not just for a better understanding of biology, but also for a better understanding of disease.”
Leading Spatialomics Platforms
There are three primary thought leaders and methodologies in the field of spatialomics. The largest of these is 10X Genomics. 10X moved into this field with two significant acquisitions, Swedish company Spatial Transcriptomics in 2018 and ReadCoor at the end of 2020. From the foundation of these acquisitions, 10X built its platform, Visium.https://www.genomeweb.com/sequencing/10x-genomics-acquire-readcoor-350m-cartana-412m#.YHyqTalKiX0 Using a spatialomics platform begins much the same as a typical tissue analysis. For Visium, users start with a tissue section and image and stain it for histological purposes. Then, the RNA is captured using bead-bound probes—barcoded for their spatial location—then sequenced. The technology can then construct the general spatial organization of gene expression. In the last step, users can analyze transcripts at the cellular level based on fluorescent in situ sequencing technology, FISSEQ. Nanostring, an emerging life science tools company, has a Spatial Molecular Imaging platform for analyzing both RNA and protein for individual cells within a tissue sample. The platform is based on the company’s Hyb & Seq chemistry technology.https://investors.nanostring.com/news/news-details/2020/NanoString-Unveils-Spatial-Molecular-Imaging-Platform-Complementing-GeoMx-DSP-to-Provide-Compelling-Product-Roadmap-for-Spatial-Biology/default.aspx

Gary Nolan, Stanford (top left) Terry Lo, CEO of Vizgen (top right), and Sean Kendall (bottom left), Keith Crandell (bottom right), both at ARCH VenturesVizgen, also a life science tools company, is the newest to this field. The company uses a molecular barcoding technology that can optically quantify single RNA molecules at a single-cell level. This is based on the MERFISH technology co-invented by Moffitt: a multiplex RNA imaging technology that can detect tens of thousands of molecules.“With non-spatial single-cell technologies, you get to see the complexity of the system but not how it fits together,” says Sean Kendall, Principal at ARCH Venture Partners and a Board Observer at Vizgen. “The Vizgen technology, for example, is subcellular, highly sensitive, and quantitative. If you want to study an individual cell or interrogate a whole pathway at the transcriptional level, you could do it. It’s faster and you can do a ton of experimentation rapidly. This gives you a level of understanding of a biological system that you were unable to get before.”Terry Lo, CEO of Vizgen, emphasized the innovation incorporated into Vizgen’s technology. “What’s been lacking in previous platforms are two things. One, the resolution to get down to the single-cell information level for gene expression. Second, sensitivity and detection efficiency. This is the ability to look at medium to low expressing genes. Oftentimes, the most biologically interesting new insights will be uncovered by these transcripts,” says Lo. “If only a small percentage of the transcripts being expressed are detected, then the full picture of what is happening with the biology of that particular cell is incomplete.”
Synthetic Biology Stands to Make Significant Discovery Gains
Spatialomics looks to be especially important for synthetic biology. As a field, synthetic biology is founded on engineering new biological systems with desirable functions. While current spatialomics research centers on biopharma, the technology could also provide new answers to key bioengineering questions in agriculture, biofuels, and other bio-based materials.Garry Nolan, Professor in the Department of Pathology at Stanford University School of Medicine, and a pioneer in single-cell analysis, and founder of Akoya, says spatialomics enables an understanding of biology at a fundamental level. “In synthetic biology, if I want to cause a tissue to organize a certain way, I better understand what the rules of the system are,” says Nolan. “So much of what we measure is hugely distant from the lowest level with the base rules of the system. Spatialomics gives us data that is closer to the fundamental thing we are interested in understanding. Until we understand these fundamental rules, we will never be good synthetic biologists.” Kendall shares Nolan’s perspective and adds the importance of spatialomics specifically to engineering new biological systems. “If you want to engineer a brain someday, you need to understand spatial context,” says Kendall. “just like a synthetic biologist might build genetic circuits at the cellular level, these tools could help build cellular networks at the tissue level.”
“A New Type of Drug Target is Coming—It’s Inevitable”
For Nolan, spatialomics paves the way for new types of disease targets. “We need to understand the rules of why certain cells, such as immune cells, come together in various disease states. We need to decipher what these observations and patterns mean,” he says.The next generation of drug targets are not going to look like today’s typical protein or enzyme drug target, says Nolan. “The targets are going to be stopping groups of cells from coming together, or enabling groups of cells to come together that wouldn’t have come together before,” Nolan says. “This new type of drug target is coming—it’s inevitable.”Walt believes that the future of spatialomics is multi-omics. This technology combines imaging with transcriptomic, proteomic, and potentially other types of analyses. Instead of focusing only on gene expression, multi-omics could create highly multiplexed images linking gene expression to other factors like protein expression, chromatin state, epigenomics, and metabolomic data.Keith Crandell, Managing Director and Co-Founder at ARCH and Board Member at Vizgen, sees the next generation technology as one of integration. He specifically cites another technology, Ultivue, a company that operates in the high-plex protein space. He sees technologies like Ultivue and Vizgen’s becoming integrated in the future.

Visualization of gene expression using MERFISH technology. Image: MERFISH“So far, we haven’t really seen that tight integration between the proteome and the transcriptome, but I think it’s coming, and then we’ll have a better idea of how much each ultimately defines a particular disease,” says Crandell. Visualizing multiple proteins at once is important as proteins are often used as key biomarkers in classifying disease. An example of this is PD1/PDL1, which is often used to identify cancers that have evolved to hide from the immune system. But, as with any emerging technology, there is always the factor of cost. For Crandell, the key driver in determining the adoption of spatialomics technology is the cost per data point at the cellular level. “I think that the critical metric is the cost per cell. And I think that's going to be subject to Emily’s Law,” says Crandell. By “Emily’s Law,” Crandell is referencing Emily Leproust, CEO of Twist Biosciences. Leproust drove down the cost of gene synthesis through Twist’s technology, making gene synthesis more accessible to researchers everywhere. “I think that’s going to happen in spatialomics.”Provided spatialomics can clear this cost hurdle, it holds particular promise in the area of neurological disease. Diseases such as Alzheimer’s and Parkinson’s have arguably the highest unmet need in medicine currently—no treatments currently exist. Paradoxically, however, potential medicines have the highest failure rate in treating these diseases.Moffitt says that this is due, in part, to our relatively rudimentary understanding of the brain compared to other organs. “What we know about the brain is that it’s very spatially organized. What we haven’t been able to do to date is understand which different cells are present and map their spatial organization,” he says. “We need to understand how those cell types are potentially changing in different biological states, and spatialomics has the ability to give us that information. I think that is what is going to lead us to an understanding of neurological diseases where we can begin to truly treat them.”Neuroscience, with all its complexities, could see significant advances through spatialomics. The technology’s potential also extends to immunology, gut microbiology, oncology, and beyond.“Spatialomics is the next evolution of genomics,” Lo says. “It’s an exciting time right now.”

Looking to hear more about how synthetic biology is changing the future of medicine? Join SynBioBeta’s special Biopharma event – Get your ticket today!
Read More
Newletter & More

SynBioBeta
Join the innovators shaping the future with SynBio + AI. From health to ag, materials & more—be part of the revolution.

















