Engineered Human Therapies
A DNA-Based Anticoagulant is Set to Disrupt the Norm
Bridging biochemistry and thrombosis biology, researchers are crafting a revolutionary anticoagulant poised to change the future of blood clot treatments
Science Photo Library (Canva)
The healthcare world is at the cusp of a monumental shift. Remember the word "anticoagulant"? If you don't, it's the medical jargon for medicines that stop blood clots, and they've been crucial in treating severe cases of heart attacks and the notorious COVID-19. Yet, here's the twist: the most popular kid on the block, heparin, doesn't always play nice. Occasionally, instead of being a hero, it makes things go south, worsening the blood clots it's supposed to prevent.
Now, imagine a world where a side-effect-free anticoagulant treatment was in sight. And no, this isn't some sci-fi novel. A team of visionary researchers, spearheaded by the University of Tokyo, is making this dream a reality. Their groundbreaking work has already shown promise in test mice, and if all goes well, we might be on the brink of human trials in a few short years.
The findings from this new study were published recently in Molecular Therapy.
Heparin's Shadows and the Lingering Legacy of COVID-19
COVID-19 wasn't just about the immediate health crisis; it unveiled gaps in our medical treatments. As the world slowly tries to heal, the ripple effects of the pandemic are still palpable. Among these is the underreported dark side of using heparin in treating severe COVID-19 cases. A tiny but significant fraction of patients—a chilling 3%—encounter a severe backlash from heparin, which ironically triggers a rapid and potentially fatal clotting of the blood, the very menace it's designed to fight.
But this isn't just a COVID-19 issue. Heart attacks, surgeries, and kidney dialysis patients have long depended on anticoagulants. Heparin, hailed by the World Health Organization and used globally, has a shadow side, especially for pregnant women. With the pharmaceutical industry turning a somewhat blind eye, there's been a gaping need for safer alternatives.
The Next-Gen Blood Savior: DNA to the Rescue
Dive deep into the laboratories at the University of Tokyo, and you'll find Associate Professor Keitaro Yoshimoto pioneering a radical solution. "The best solution would be to sidestep HIT (heparin-induced thrombocytopenia) altogether," says Yoshimoto. "But since that's still a dream, our focus has been on innovating a low-risk thrombin inhibitor."
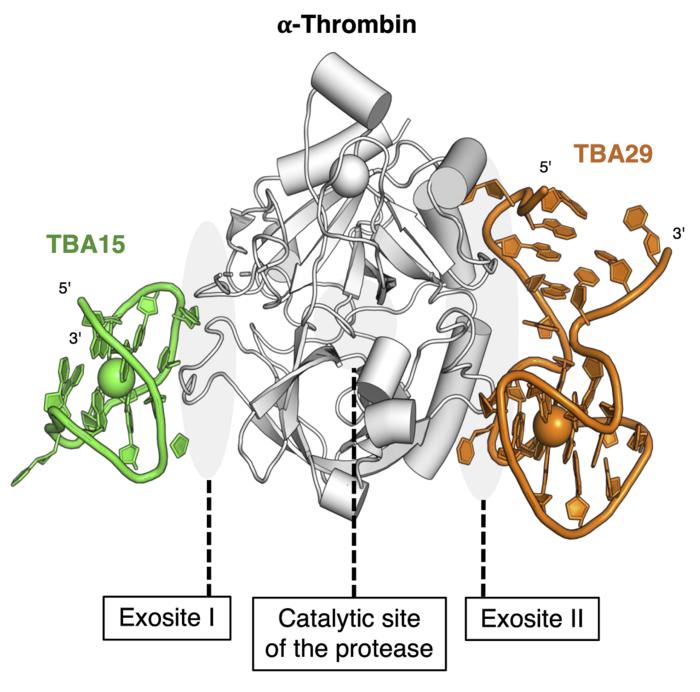
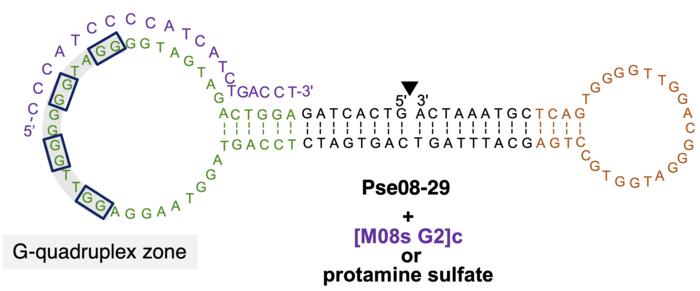
Yoshimoto and his stellar team have crafted an anticoagulant that’s not just any drug—it's DNA-based. This next-gen thrombin inhibitor is a blend of DNA molecules armed with the power to prevent severe bleeding. Its secret weapon? A molecule known as a bispecific aptamer can multitask by binding to multiple targets simultaneously. Moreover, the DNA segments in this drug are like a safety net, ready to swoop in if undesirable clotting threatens to emerge.
The Promise and the Road Ahead
Steered by Yoshimoto's expertise in biochemistry and molecular separation and in collaboration with Assistant Professor Asuka Sakata, a maestro in thrombosis biology, this revolutionary treatment is on the fast track. Yoshimoto hints at the future, saying, "We're gearing up for human trials. The journey ahead includes two years of preclinical studies and five for human trials. Even if HIT impacts a small number, its severity demands our immediate attention."
In a nutshell, we're witnessing the dawn of a transformative era in blood clot treatments—one that sidesteps traditional chemistry, harnesses the potential of DNA, and promises a brighter, safer future for patients worldwide.