Biomanufacturing Scale Up
Harnessing Biofilms for Biomanufacturing and as Living Materials
The resilience of microbial biofilms suits their use in chemical production and as advanced materials
[Science Photo Library/Canva]
Early in the history of life on Earth, microbes learned a neat trick. They aggregated on surfaces into clusters to either capture new niches or improve survival. Most bacteria and many microalgae, fungi, and archaea can produce these structures called biofilms. And they are incredibly robust.
“That’s what makes them difficult to combat when they are not wanted,“ said Katja Bühler, a Helmholtz Centre for Environmental Research microbiologist. For example, bacterial biofilms growing on medical devices are a major cause of hospital-related infections. However, “in biotechnology, this feature can be turned into an advantage,” said Bühler.
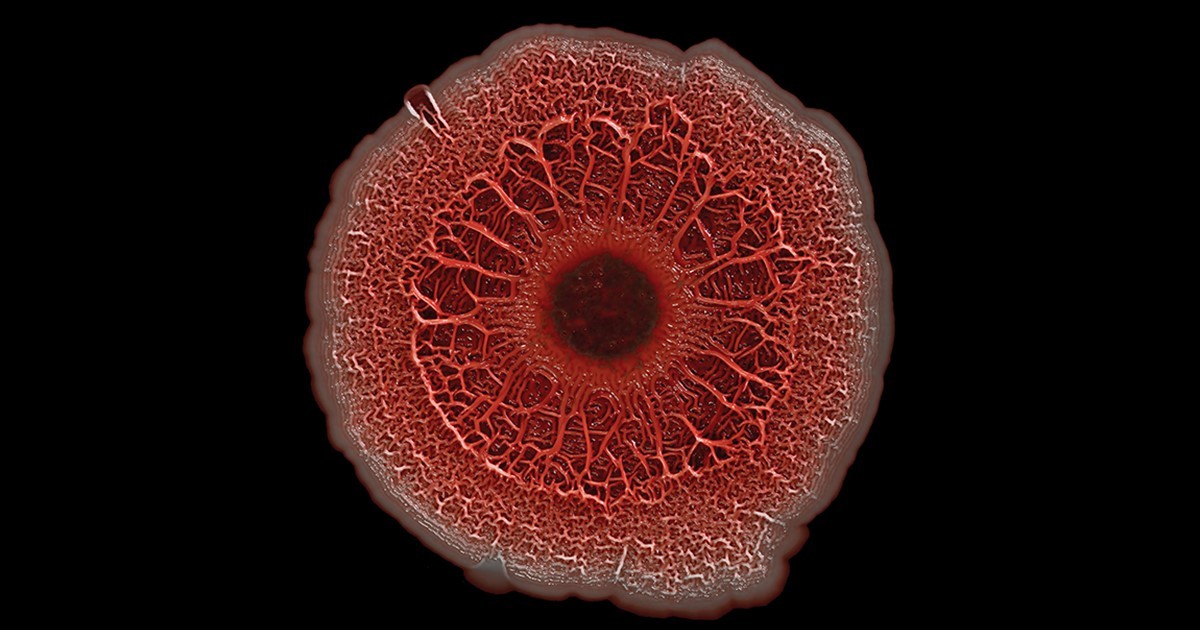
Producing Materials in Biofilm Reactors
Biofilms evolved to be biologically robust. “Microbes within a biofilm can survive exposure to many harsh chemicals that would kill them if encountered outside of a biofilm,” said Anne Meyer, a University of Rochester synthetic biologist. Speaking on their physical robustness, Meyer added that “they have high viscoelasticity and adhere tightly to physical substrates so that they can withstand many types of physical insults.”
Moreover, microbial biofilms' intrinsic self-healing nature helps them maintain their activities over extended periods. Combined with their ease of attachment on surfaces, this makes them suitable for lining the walls of continuous bioprocess reactors. Biofilm reactors that routinely find use in wastewater treatment work on this principle.
Now, with catalytic biofilms, similar reactors are finding use in biomanufacturing. The benefits of biofilms extend beyond their robustness. "Biofilms and pellicles are the states when microbes produce a material in any significantly measurable amounts,” said Tom Ellis, a synthetic biologist at Imperial College London. “Some microbes, when they are in a biofilm state, have increased carbon flux into carbon storage pathways,” said Andrew Magyar, CTO of US biotech Capra Biosciences.

The company develops a continuous fermentation reactor that upcycles food and agricultural waste into fat-soluble chemicals like retinol and ester-based lubricants. Its reactor is lined with the biofilm-forming bacterium Marinobacter atlanticus. Capra Biosciences CEO Elizabeth Onderko will be speaking about biomanufacturing at this year's Global Synthetic Biology Conference in a session entitled “SynBio-Enhanced Supply Chain Security: Feedstocks & Distributed Manufacturing for Climate Resilience.”
Some materials are easier to produce in biofilm bioreactors. “Biofilms are well suited for all reactions involving secreted products, particularly gaseous ones,” said Bühler. On the contrary, Bühler added intracellular products that require a cell lysis step for purification are better produced in conventional bioreactors.
Capra’s technology does not involve the lysis step either. “We use a solvent that is immiscible with water and does not harm our microbes to remove the product. This allows in situ product extraction without needing to harvest the cells,” said Magyar.
Biofilms as Advanced Materials
The structural distinctness of biofilms also enables engineering specific material properties. For example, “biofilms are very efficient at producing polymer networks that are tightly interwoven,” said Meyer. This is why, beyond leveraging them to produce materials, companies are developing biofilms as advanced materials.
For example, UK-based biotech Modern Synthesis creates a biomaterial fabric that is part biofilm and part textile. The company “takes microbially-grown bacterial nanocellulose and uses material science to form a customized biofilm, which is then combined with a textile element to form a finished composite material,” said CTO and co-founder Ben Reeve. The company’s CEO, Jen Keane, will also be at this year's Global Synthetic Biology Conference in a session entitled “The New Biomaterials Innovators.”
The biofilm component of the biomaterial fabric is tunable to meet specific material requirements. “If a product application requires our material to hit a particular stiffness or stretch value, we can precisely control both of those parameters to deliver to a brand’s requirements,” said Reeve.
In recent years, biomaterials have been reimagined as responsive or living materials and not just inert materials of biological origin. Scientists are fabricating engineered living materials at different scales. There are engineered biofilms as well as living materials built with biofilm-producing microbes or polymer components of biofilms, such as bacterial cellulose.
“We use synthetic biology to engineer material over-producing cells, like Komagataeibacter rhaeticus, to also produce other functional elements like enzymes or chemical sensors,” said Ellis. “The grown material can have functional properties or be a living material that holds cells within it that sense and respond to a chemical.”
Ellis and Modern Synthesis collaborated to engineer K. rhaeticus bacteria to produce black pigment as they produce nanocellulose. “This innovation resulted in self-dyeing materials that can be used as more responsible alternatives to leather,” said Reeve. The bacteria produced a stable color, and “this pigment can also be produced in specific patterns in response to stimuli such as light or heat”, added Reeve.

While still early, scientists are exploring applications of engineered biofilms. Ellis said that researchers are creating strong glues with biofilms, for instance. “These can be used to fix cracks or to coat boat hull surfaces to prevent algal fouling and build-up of barnacles,” added Ellis.
Engineered biofilms could have applications in wound healing and tissue scaffolding as well. “Biofilms can be cultivated with biocompatible polymers, forming hydrogels that can encapsulate and stabilize delicate biological molecules.” This could enhance tissue regeneration and combat infections more effectively.
What’s Next?
Producing materials in biofilm reactors and biofilms as living materials are both young and exciting themes at the intersection of biology and materials science. As they mature, they will benefit from an exchange of insights.
For instance, the know-how of biofilm reactors could help develop biofilms as functional materials. The pipeline for the latter “would be more efficient with better tools for growing different types of biofilms in parallel at medium- and large-scale,” said Meyer. These also need to be “supplied with nutrients on the longer timescales that biofilms typically need to mature and develop their emergent biological and physical resistance properties”.
In the other direction, engineered biofilms could improve functionality and control in biomanufacturing. Incorporating new functionalities to biofilm-producing strains via genetic engineering and new ways to hybridize biofilms with other materials will pave the way for more advanced biofilm-based materials.
However, there's still a lot of fundamental work to be done. “From a scientific standpoint, an area that has the potential to further advance technologies like ours is gaining a better understanding of the genetic basis for biofilm formation,” said Magyar.
Read More
Newletter & More

SynBioBeta
Join the innovators shaping the future with SynBio + AI. From health to ag, materials & more—be part of the revolution.