Ai Digital Biology
Next-Gen Biosensors Light the Way on Disease Detection
A new synthetic biology platform promises to revolutionize biosensors, enabling faster, more accurate disease detection

[ideogram.ai]
Biosensors, devices that leverage biological molecules to identify the presence of target substances, are poised to revolutionize fields from disease diagnostics to environmental monitoring. Among these, fluorescent biosensors are widely recognized for their potential to detect disease biomarkers, biological processes, and environmental toxins. Yet, traditional fluorescent biosensors face a significant challenge: their fluorescent probes are often "always on," leading to low-contrast signals and requiring complex washing steps to differentiate between bound and unbound molecules.
Enter high-contrast binding-activated fluorescent biosensors—innovative nanosensors that illuminate only when bound to their target molecule. Despite the breakthrough potential, engineering these nanosensors has been difficult, demanding the integration of effective target binding and an on-demand fluorescence switch in a compact, scalable design.
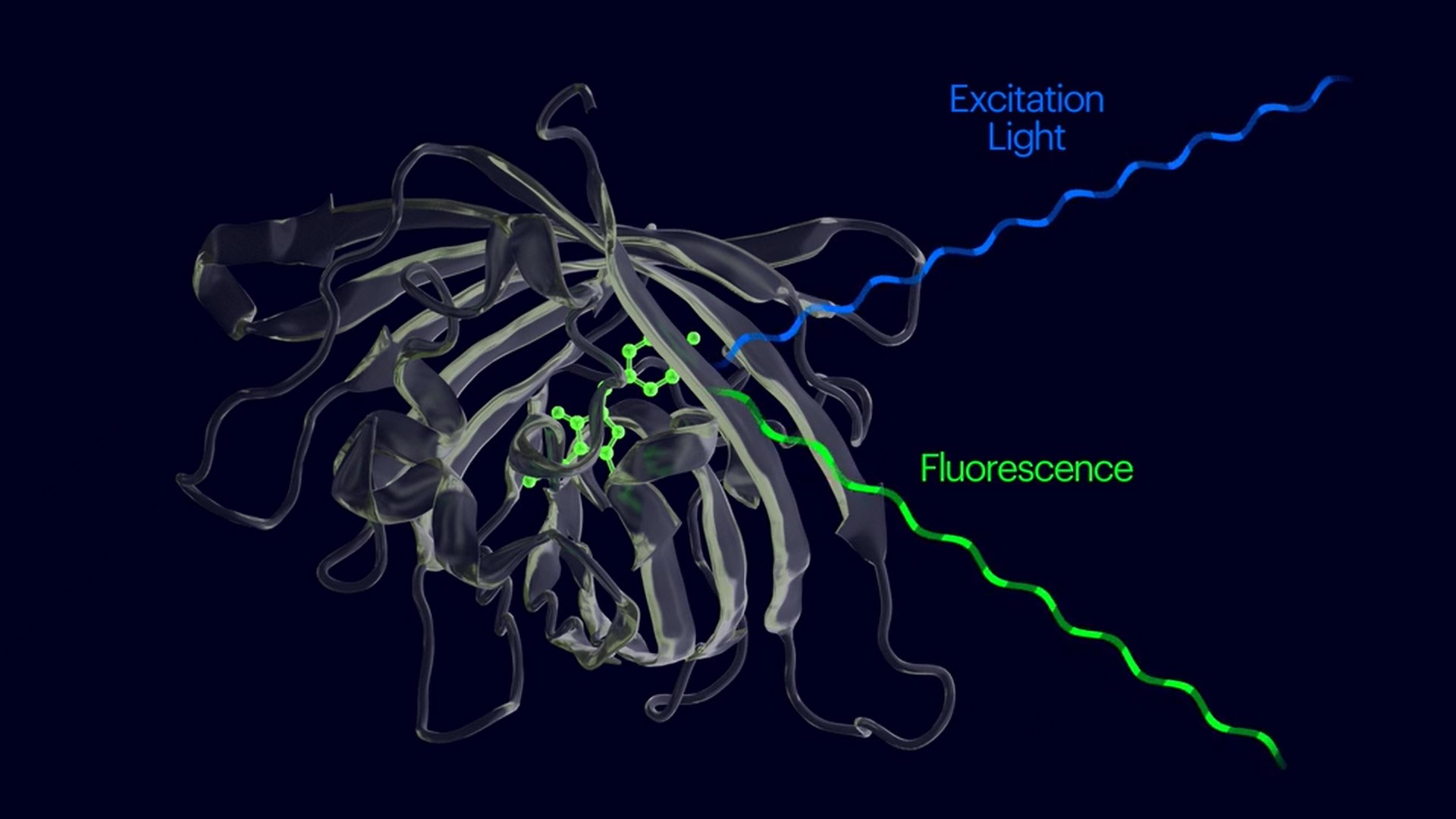
A team of researchers from the Wyss Institute at Harvard University, Harvard Medical School (HMS), MIT, and the University of Edinburgh has now developed a cutting-edge synthetic biology platform that transforms how nanosensors are discovered, evolved, and manufactured. Their breakthrough enables nanosensors to detect specific proteins, peptides, and small molecules with a 100-fold increase in fluorescence in less than a second. Central to this innovation are fluorogenic amino acids (FgAAs), new building blocks that are encoded into small protein binders using an advanced genetic expansion methodology. This platform paves the way for a wide array of applications, from environmental monitoring to medical diagnostics, and is already yielding exciting results.
A Synthetic Biology Breakthrough
"We've been working on expanding the genetic code for years, aiming to give cells new capabilities for research and medicine. This new platform is a significant step forward," said George Church, PhD, leader of the study and a prominent figure at the Wyss Institute. The platform, outlined in Nature Communications, offers a powerful solution to the long-standing challenges of enhancing proteins with new chemistries—creating an instant, highly capable biosensor platform with transformative implications for biotechnology.
The team, led by Erkin Kuru, PhD, built on their previous discovery that FgAAs can be used to turn known protein binders into optical sensors that light up only when they bind their target. Collaborating with Marc Vendrell, PhD, a biomedical imaging expert at the University of Edinburgh, Kuru and the team initially focused on developing an "instant COVID-19 diagnostic" by engineering a miniature antibody (nanobody) that binds to the SARS-CoV-2 Spike protein. Through a series of targeted mutations and chemical modifications, they created hundreds of variants of the nanobody, selecting the fluorogenic versions that produced the strongest fluorescent signals upon binding.
This animation explains the engineering of binding-activated biosensors and how they could be applied. [Wyss Institute at Harvard University]
The platform's flexibility allowed them to extend this process to develop nanosensors for other key targets, including cancer markers and stress hormones. The nanosensors proved effective not only in test tubes but also in living human cells and bacteria, demonstrating their utility as powerful imaging tools for real-time biological studies.
Accelerating Nanosensor Evolution
The team's first version of the platform, while groundbreaking, was labor-intensive and time-consuming. "We wanted to push the limits of molecular design, so we created a more high-throughput version," said Kuru. By engineering the ribosome—the cellular machinery responsible for protein synthesis—the team developed a cell-free process that allowed them to produce millions of nanosensor candidates simultaneously.
A key innovation was the introduction of a synthetic amino acid with a pre-attached fluorogenic scaffold. This advance eliminated the need for complex post-production steps, enabling the researchers to rapidly test hundreds of nanosensor variants in a single day. Using this approach, they discovered nanosensors with superior binding affinities for the SARS-CoV-2 Spike protein, even identifying variants capable of detecting newer Omicron strains.
Looking ahead, the team envisions broad applications for their platform. "This is a major leap forward in designing cost-effective fluorescent biosensors with huge potential for diagnostics and precision medicine," said Vendrell. "We can also use this technology to create new therapeutics and research tools," added Kuru, whose team is now collaborating on the Wyss Institute’s AminoX project to develop advanced therapies based on this platform.
The Future of Biosensing
This next-generation platform represents a paradigm shift in how fluorescent biosensors are developed and deployed. "It's a perfect example of the power of synthetic biology," said Donald Ingber, MD, PhD, Founding Director of the Wyss Institute. By reengineering the fundamental processes of protein synthesis, the team has unlocked a versatile platform with vast potential to address pressing challenges in diagnostics and therapy.
With its rapid, cost-effective, and scalable approach, the new platform is set to revolutionize disease monitoring and treatment—transforming not only biosensing but the broader fields of biotechnology and precision medicine.
Read More
Newletter & More

SynBioBeta
Join the innovators shaping the future with SynBio + AI. From health to ag, materials & more—be part of the revolution.